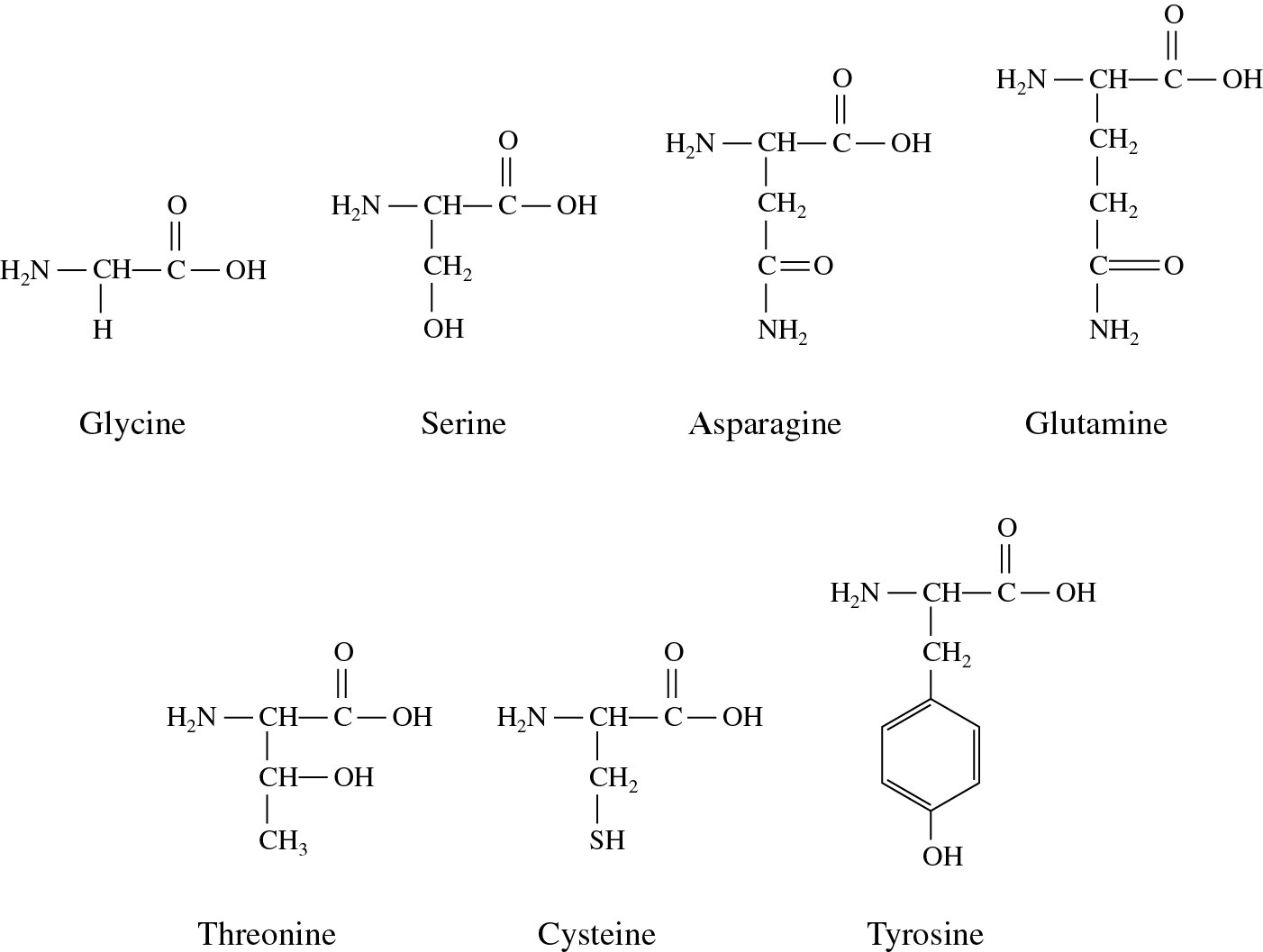
Here we focus on one factor contributing to the hydrophobic effect: solute–water interactions, for simplicity referred to as solute hydrophobicity. Still, a caveat of using fluorination to control protein properties remains: do we understand the factors influencing the hydrophobic effect involving fluorinated amino acids? The answer is, simply, that we do not. Abbreviations for fluorinated amino acids follow a three-character nomenclature: initial character of parent (non-fluorinated) amino acid name (E, P, V, I, L) number of fluorine atoms (1, 2, 3, 4, 6) fluorination site (δ carbon as D, γ carbon as G or, in the case of chiral center formation following fluorination, R or S). Each amino acid residue is capped at the N-terminus with an acetate group (ACE, –COCH 3) and at the C-terminus with an N-methyl group (NME, –NHCH 3). 1 Molecular structures, commonly used names and abbreviations for the amino acids under study. 3,16–19 Even fluorinating only a few residues may enhance the hydrophobicity and passive diffusion of peptides through membranes, 20 the proteolytic resistance 21 and anti-microbial activity of proteins, 22 in addition to tuning their thermal stability, 23 making this synthetic approach of wide interest. 1), can solve this problem while simultaneously enhancing other properties of interest. those where hydrogen atoms in side chain groups are substituted by fluorine (see Fig. Fluorinated versions of those amino acids, i.e. 14,15 This is, however, difficult with the limited pool of canonical hydrophobic amino acids because their side chains differ in structure and volume. 9–13 Simultaneously, minimizing changes in solute–solute packing upon mutations is desirable to ensure that protein structure – and thus function – is preserved.
#Hydrophobic amino acids polarity how to#
7,8 Understanding how to use amino acid mutations to control the hydrophobic effect is critical to develop new protein-based drugs, biodevices and materials. 5,6 The hydrophobic effect reflects the balance between solute–water interactions and direct, predominantly dispersive, solute–solute interactions. 1 Introduction The preferential interaction between apolar solutes in water – the hydrophobic effect – is a key factor driving protein folding, 1,2 structural stability with respect to changes in temperature 3,4 (thermal stability) and interactions with other proteins and ligands.

Our results offer a road map to mechanistically understand how fluorination alters hydrophobicity of (bio)polymers.
Changes in Δ G Hyd reflect two main contributions: (i) fluorination alters side chain–water interactions we identify a crossover point from hydrophilic to hydrophobic fluoromethyl groups which may be used to estimate the hydrophobicity of fluorinated alkyl side-chains (ii) fluorination alters the number of backbone–water hydrogen bonds via changes in the relative side chain-backbone conformation. Fluorination changes Δ G Hyd by −1.5 to +2 kcal mol −1, but the number of fluorines is a poor predictor of hydrophobicity.
#Hydrophobic amino acids polarity free#
We use molecular dynamics simulations, together with a new fixed-charge, atomistic force field, to quantify the changes in hydration free energy, Δ G Hyd, for amino acids with alkyl side chains and with 1 to 6 –CH → –CF side chain substitutions. Key to the impact of fluorination on protein properties is the hydrophobicity of fluorinated amino acids. Fluorination can dramatically improve the thermal and proteolytic stability of proteins and their enzymatic activity.
